There has been some concern regarding the statistics periodically issued by the Patent Trial and Appeal Board (PTAB) of the US Patent and Trademark Office, that the numbers reported overlook multiple inter partes review (IPR) challenges to the same patents, and potentially, different outcomes in those challenges. We have monitored IPRs filed on drug patents – patents that are listed in the Food and Drug Administration's (FDA) Orange Book ("Orange Book patents"), and patents that have been identified in proceedings as reading on FDA Purple Book listed biologic drugs (Biologic Drug Patents) – and report here that while certain drug patents have been challenged in multiple IPR petitions, concern as to different outcomes, at least in the final written decisions (FWDs) that have been issued to date, appears to be unfounded. These FWDs have been consistent: either all instituted claims have been held unpatentable, or all instituted claims have been held not unpatentable. The biggest news is that drug patents challenged in multiple IPRs – at least those that reach FWD – have a much greater chance of being found unpatentable than drug patents that have been challenged in only one IPR. The percentage is 63% for what we have described in this article as "duplicative FWDs", compared to 44% for non-duplicative FWDs.
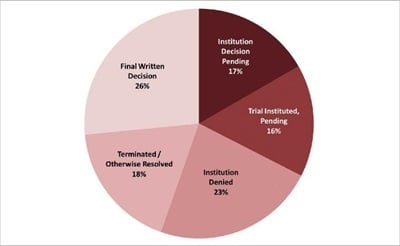 As of 20 July 2017, there have been more than 350 IPR petitions filed against Orange Book patents, and more than 70 IPRs petitions filed against Biologic Drug Patents. Of these drug patent IPR petitions, 67% have been resolved (institution has been denied in 23%; a FWD has issued in 26%; and IPR has otherwise been terminated, for example, following settlement, in 18%); 16% have been instituted and trial is pending; and the remaining 17% are awaiting an institution decision (see figure 1). This article focuses on the subset of drug patent IPRs that have been resolved, and in particular on those FWDs that concern drug patents that were also the subject of at least one other FWD, which we describe as "duplicative FWDs".
As of 20 July 2017, there have been more than 350 IPR petitions filed against Orange Book patents, and more than 70 IPRs petitions filed against Biologic Drug Patents. Of these drug patent IPR petitions, 67% have been resolved (institution has been denied in 23%; a FWD has issued in 26%; and IPR has otherwise been terminated, for example, following settlement, in 18%); 16% have been instituted and trial is pending; and the remaining 17% are awaiting an institution decision (see figure 1). This article focuses on the subset of drug patent IPRs that have been resolved, and in particular on those FWDs that concern drug patents that were also the subject of at least one other FWD, which we describe as "duplicative FWDs".
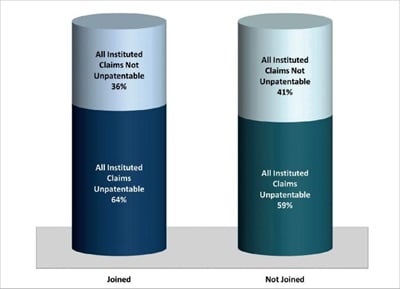 As of 20 July 2017, from the more than 420 IPR petitions filed to date against drug patents, there have been at least 72 duplicative FWDs. Of these 72 duplicative FWDs, 55 (76%) were on joined IPRs.1 The remaining 17 FWDs were separate decisions that issued on IPRs that had not been joined. Looking first at the joined IPRs that reached FWD: 35 (64%) resulted in FWDs in which all instituted claims were unpatentable; and the remaining 20 (36%) resulted in FWDs in which all instituted claims were not unpatentable. Turning next to duplicative FWDs that were not joined: the PTAB found that in 10 (59%) all instituted claims were unpatentable; and in the remaining 7 (41%) all instituted claims were not unpatentable, (see figure 2). In other words, slightly more challenged drug patents lost claims when duplicative IPRs were not joined, but the difference was minimal.
As of 20 July 2017, from the more than 420 IPR petitions filed to date against drug patents, there have been at least 72 duplicative FWDs. Of these 72 duplicative FWDs, 55 (76%) were on joined IPRs.1 The remaining 17 FWDs were separate decisions that issued on IPRs that had not been joined. Looking first at the joined IPRs that reached FWD: 35 (64%) resulted in FWDs in which all instituted claims were unpatentable; and the remaining 20 (36%) resulted in FWDs in which all instituted claims were not unpatentable. Turning next to duplicative FWDs that were not joined: the PTAB found that in 10 (59%) all instituted claims were unpatentable; and in the remaining 7 (41%) all instituted claims were not unpatentable, (see figure 2). In other words, slightly more challenged drug patents lost claims when duplicative IPRs were not joined, but the difference was minimal.
The biggest difference can be seen when the statistics for these duplicative FWDs are compared with those for FWDs issued on drug patents that have to date only been challenged in one IPR resulting in a FWD ("single FWDs"). The difference in these numbers is much more significant. In total, combining all duplicative FWDs on drug patents as of 20 July 2017 (joined and not joined): the PTAB found all instituted claims unpatentable in 45 (63%) of these FWDs; and all instituted claims not unpatentable in 27 (38%) of these FWDs.
 Comparing this to the same metrics for single FWDs on drug patents: in single FWDs, the PTAB found all instituted claims unpatentable in 19 (43%) FWDs; some instituted claims patentable and some unpatentable in two (5%) FWDs; and all instituted claims not unpatentable in 23 (52%) FWDs, (see figure 3). In other words, challenged claims of drug patents are much more likely to be found unpatentable when there have been multiple versus single FWDs on the same patent – 63% versus 43% – although this may be a product of the strength of the patents themselves as opposed to a "second bite of the apple" being more successful.
Comparing this to the same metrics for single FWDs on drug patents: in single FWDs, the PTAB found all instituted claims unpatentable in 19 (43%) FWDs; some instituted claims patentable and some unpatentable in two (5%) FWDs; and all instituted claims not unpatentable in 23 (52%) FWDs, (see figure 3). In other words, challenged claims of drug patents are much more likely to be found unpatentable when there have been multiple versus single FWDs on the same patent – 63% versus 43% – although this may be a product of the strength of the patents themselves as opposed to a "second bite of the apple" being more successful.
There are a number of observations that can be made, and lessons that can be learned from these duplicative FWDs. First, even outside joinder (where a party seeks to join a petition), at least to date, the PTAB appears to be coordinating, and is issuing similar FWDs on these IPRs, even when the oral hearings are held and decisions are issued months apart. Secondly, thus far, all of these duplicative FWDs have assessed the patentability of instituted claims on the ground of obviousness, and all have assessed the patentability of instituted claims based on at least one prior art reference cited during prosecution. Thirdly, despite criticism that the PTAB has a tendency to give short shrift to secondary considerations, where a prima facie case of obviousness was found, the PTAB included in its analysis consideration of objective evidence of non-obviousness (including unexpected results, long-felt but unmet need, industry praise and commercial success). Where this evidence was deemed unsuccessful in tipping the balance in the patent owner's favour, the PTAB generally reasoned that this was because of insufficient evidence of a nexus between the objective evidence, and the claimed invention. There has been at least one, very notable exception to this: the Prolensa FWDs (IPR2015-00902 and -01099).
Prolensa FWDs
In the Prolensa FWDs, the PTAB was persuaded, that on the preponderance of the evidence, objective evidence of unexpected results, long-felt need, and commercial success, presented by the patent owner, outweighed the petitioners' prima facie case.
The Prolensa IPRs concerned challenges to all 30 claims of US patent No 8,669,290 (the '290 patent), owned by Senju Pharmaceutical (Senju), as obvious based on one prior art patent listed on the face of the '290 patent, and one new prior art patent that was not cited during prosecution. The '290 patent concerns aqueous liquid preparations for ophthalmic administration, consisting of two components: bromfenac (or its salts or hydrates) (a non-steroidal anti-inflammatory drug (NSAID)), and tyloxapol (which stabilises the bromfenac component). The PTAB found that the prior art patent listed on the face of the '290 patent disclosed every limitation of claim 1 of the '290 patent, except that it disclosed use of polysorbate 80, instead of a specific concentration of tyloxapol. This limitation was satisfied by the new prior art patent, which disclosed an aqueous ophthalmic preparation of a different NSAID, diclofenac potassium salt, and tyloxapol, in an amount that fell within the claimed concentration range. Based on the record presented at the oral hearings held in April and June of 2016, the PTAB found that a person of ordinary skill in the art would have known that polysorbate 80 and tyloxapol could be substituted successfully and predictably, because these compounds had previously been used interchangeably in ophthalmic formulations. The PTAB also found that this known interchangeability was sufficient to establish a prima facie case of obviousness, even in the absence of an express suggestion to use tyloxapol. The PTAB then shifted its attention to the patent owner Senju's evidence of unexpected results, commercial success, and industry praise. Based on the record, the PTAB found, among other things, that: (1) substituting polysorbate 80 for tyloxapol – including, unexpectedly and counter-intuitively, a reduced concentration of tyloxapol – had the surprising and unexpected result of a significant improvement in the stability of bromfenac; (2) the use of tyloxapol lowered the pH to close to that of natural tears, and meant that amounts of other irritating ingredients could also be reduced; (3) the claimed inventions were embodied in Prolensa, a product that is commercially successful at least in part because it lacked the burning and stinging side effects of other treatments; and (4) Prolensa had received significant industry acclaim based on benefits flowing from the use of tyloxapol. In short, the Prolensa IPRs are a patent owner's success story: they show that strong, robust evidence of objective indicia of non-obviousness may be enough to overcome a prima facie case of obviousness, even in IPR, and even in cases where the patent at issue has been challenged in subsequent IPR petitions.
At the other end of the spectrum are the recently issued Humira FWDs (IPR2016-00172, -00408 and -00409). These Humira IPRs concerned challenges to all five claims of US patent No 8,889,135 (the '135 patent), owned by AbbVie Biotechnology (AbbVie). Petitioners Coherus Biosciences and various Boehringer Ingelheim companies challenged these claims as obvious based on various combinations of prior art references, all of which were cited during prosecution of the '135 patent.
The '135 patent claims methods for treating rheumatoid arthritis by subcutaneously administering an anti-tumor necrosis factor α (anti-TNFα) antibody, which has the six complementary determining regions (CDRs) and heavy chain constant region of D2E7 (a recombinant human anti-TNFα antibody), in a specific dosage regimen (40mg once every 13-15 days). The PTAB found that, collectively, the cited prior art disclosed each and every element of all of claims 1-5, and there was a motivation to combine the various teachings, with a reasonable expectation of success. As to objective evidence of non-obviousness, AbbVie argued unexpected results, long-felt unmet need and commercial success. But the PTAB was not persuaded: it found that (1) there was insufficient evidence that the efficacy of the claimed invention would have been unexpected; (2) long-felt need may instead have been satisfied by the introduction of the first fully human anti-TNFα antibody; and (3) while the commercial embodiment of the claimed inventions, Humira, was commercially successful, it was not clear from the evidence presented whether there was a nexus to the claimed inventions, or whether, for example, commercial success was a result of the formulation, or the result of known and patented fully human D2E7 antibody. In three FWDs issued in May and July 2017, the PTAB found all five challenged claims unpatentable.2
Summary
The biggest takeaways from our analysis is that, while multiple IPRs challenging claims of the same drug patent increase the risk that claims will be found unpatentable, at least so far, the PTAB does appear to be coordinating these duplicative IPRs, even outside joinder. Also, patent owners should not lose hope as to secondary considerations: the Prolensa IPRs are a testament that robust objective evidence of non-obviousness can carry the day. In any event, patent owners must take care to take consistent positions, across their patent portfolios, when responding to multiple challenges.
- The number of individual FWDs issued was 24, because each FWD ruled on at least two joined IPR proceedings.
- Patent owner AbbVie filed notices of appeal to the Federal Circuit concerning IPR2016-00172 on 14 July 2017 (Fed Cir 17-2304), and concerning IPR2016-00408 and -00496 on 31 July 2017 (Fed Cir 17-2362 and 17-2363).